Bacterial Vaginosis (BV) occurs when normal hydrogen peroxide producing Lactobacillus sp. is replaced by an overgrowth of facultative anaerobic bacteria. If a woman presents with symptoms, including vaginal discharge, irritation and malodor
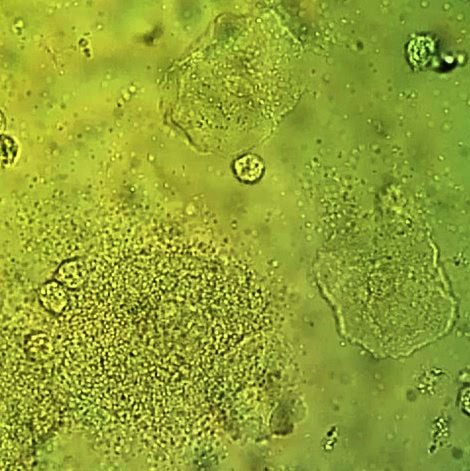
Vaginal wet mount with a NaCl preparation, showing a clue cell at bottom left, and two normal epithelial cells.
Bacterial Vaginosis (BV) is not caused by a single bacterium, but by high concentrations of facultative anaerobic bacteria (e.g., Prevotella sp. and Mobiluncus sp.), G. vaginalis, Ureaplasma, Mycoplasma, and numerous other anaerobes. BV is associated with multiple or new sex partners, lack of condom use and in particular lack of vaginal lactobacillus. Because BV is not an inflammatory condition, vulvar erythema and edema are not commonly seen, unlike candidiasis and trichomoniasis.
Treatment is recommended for women with symptoms, including discharge, irritation and malodor and may reduce the risk for C. trachomatis, N. gonorrhoeae, T. vaginalis, HIV, and herpes simplex type 2.
Recommended CDC regimens include the following:
Alternative Regimens
*Alcohol consumption should be avoided during treatment with oral nitroimidazoles. To reduce the possibility of a disulfiram-like reaction, abstinence from alcohol use should continue for 24 hours after completion of metronidazole and 72 hours after completion of tinidazole.
**Clindamycin ovules use an oleaginous base that might weaken latex or rubber products (e.g., condoms and vaginal contraceptive diaphragms). Use of such products within 72 hours following treatment with clindamycin ovules is not recommended.
The USPSTF recommends against screening for bacterial vaginosis in pregnant persons not at increased risk for preterm delivery. (D recommendation)
The USPSTF concludes that the current evidence is insufficient to assess the balance of benefits and harms of screening for bacterial vaginosis in pregnant persons at increased risk for preterm delivery. (I statement)
Sexually Transmitted Infections Treatment Guidelines 2021
ACOG Practice Bulletin 215: Vaginitis in Nonpregnant Patients
USPSTF: Screening for Bacterial Vaginosis in Pregnant Persons to Prevent Preterm Delivery
Are you an
ObG Insider?
Get specially curated clinical summaries delivered to your inbox every week for free
Please log in to ObGFirst to access this page
OBG Project CME requires a modern web browser (Internet Explorer 10+, Mozilla Firefox, Apple Safari, Google Chrome, Microsoft Edge). Certain educational activities may require additional software to view multimedia, presentation, or printable versions of their content. These activities will be marked as such and will provide links to the required software. That software may be: Adobe Flash, Apple QuickTime, Adobe Acrobat, Microsoft PowerPoint, Windows Media Player, or Real Networks Real One Player.
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The planners of this activity do not recommend the use of any agent outside of the labeled indications.
The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of the planners. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information
presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patient’s conditions and possible contraindications and/or dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
It appears you don't have enough CME Hours to take this Post-Test. We no longer offer Hours.
You are now leaving the ObG website and on your way to PRIORITY at UCSF, an independent website. Therefore, we are not responsible for the content or availability of this site
