Male Factor Infertility and Interpreting the Semen Analysis
In about half of couples experiencing infertility, there is a male factor which either contributes to, or is responsible for, their subfertility. This common condition presents a challenge to Reproductive Endocrinology and Infertility (REI) professionals, as it is a complicated diagnosis with many causes and treatments and our formal education on the topic is, arguably, limited. This lack of information (combined with the reduced visibility and familiarity of the male partner in the infertility setting) can result in a healthcare professional experiencing discomfort when discussing results and treatment options. Semen analysis remains the primary test for male fertility, and health care providers are often the ones who interpret and discuss these results with patients. The goal of this article is to review the sperm cell itself, its journey through male and female reproductive tracts, and how to interpret the semen analysis properly. The psychosocial aspects of male infertility will also be reviewed in an effort to improve provider/patient communication when relaying results.
Accompanying this article is the following video from Fertile Health, LLC.
The Sperm and Sperm Journey
The sperm cell has one main role: to transfer paternal DNA by reaching, attaching to, penetrating and activating the female oocyte and its shape is highly adaptive to its function. A sperm cells consist of four main segments: The head (which contains the nucleus and the acrosome, an organelle which is critical for penetrating the zona pellucida of the oocyte); the neck (which contains centrioles); the midpiece (which contains the mitochondria, the energy provider); and the tail (which has flagellum that propels the sperm). Sperm cells are unique in that they mostly lack cytoplasm, which is extruded as they differentiate from round spermatids to mature spermatozoa with the typical shape with which we are familiar: a small oval head (mostly consisting of the nucleus) and a long tail. So, basically, a sperm cell is a small, energetic torpedo that delivers male genetic material. Sperm cells are produced approximately every 46-72 days by a complex process called spermatogenesis which occurs in the seminiferous tubules of the testes. Spermatogenesis is affected by intrinsic factors, such as a properly functioning hypothalamic-pituitary-testicular axis, and extrinsic factors, such as heat. In fact, the testes reside outside of the body in order to provide thermoregulation (they are 2-3 degrees cooler than rectal temperature). Once spermatogenesis is complete, the sperm enter the epididymis, a comma-like structure on the outside of the testicle, where they finish the maturation process and become motile. They remain in the epididymis for 2-3 months or until the time of ejaculation, which is an important point as sperm that is ejaculated was produced at least 2-3 months prior.
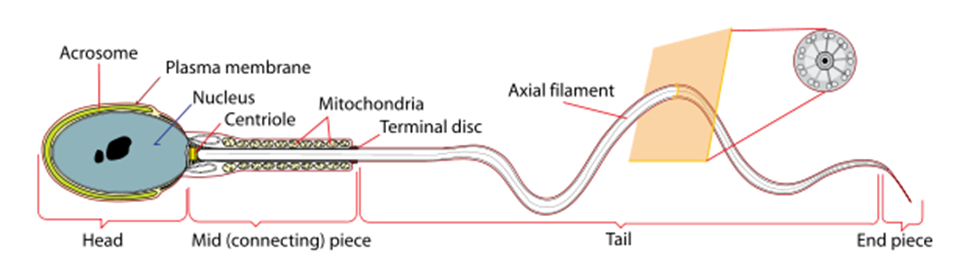
(Image credit: Mariana Ruiz Villarreal)
In order to travel through the female reproductive tract, sperm needs to combine with fluid in which it can move. This fluid is generated in the male by several structures: the seminal vesicles, the prostate gland and urethral glands and, once combined, called seminal fluid or semen. Semen also provides fructose, the energy source necessary for sperm movement, and other substances that protect sperm during its journey through the reproductive tract, as sperm cells are vulnerable to a myriad of external insults, such as temperature changes, acidic pH and oxidative stress.
The sperm journey through the female reproductive tract closely resembles an obstacle course, fraught with barriers and challenges, and a healthy sperm must be equipped to overcome them. Once ejaculated, the seminal fluid provides substances that protect the sperm and assist it during its journey, such as agents which buffer the acidic environment of the vagina, substances that assist with motility through cervical mucus and still others which suppress the immune response of the female which can destroy sperm. Those sperm which survive to enter the fallopian tubes remain there for a period of time where the glycoprotein coating is removed from the surface of the sperm (called capacitation), a necessary process where sperm acquire the ability to fertilize an egg. The acrosome, the organelle covering the head of the sperm, is filled with the enzymes necessary to penetrate the zone pellucida of the oocyte, allowing one sperm to enter and fertilize and activate the egg. The remaining sperm die off in a process called apoptosis. There is a huge attrition rate from the sperm present in the ejaculate to those that reach the zone pellucida, so in order to account for this, many millions of motile sperm need to be deposited in the vagina at the time of intercourse in order for just tens of hundreds to reach the ampulla of the fallopian tubes, where fertilization usually occurs.
Diagnosis of Male Infertility
A comprehensive evaluation of the male has three necessary components: A thorough medical history (including a sexual and reproductive history), a physical exam, and a semen analysis, the main diagnostic test for male factor infertility. There seems to be agreement among most experts that the initial evaluation should include a medical history and a semen analysis and, if abnormal, then the patient should be referred to a male reproductive specialist with the expertise and experience to complete the physical exam.
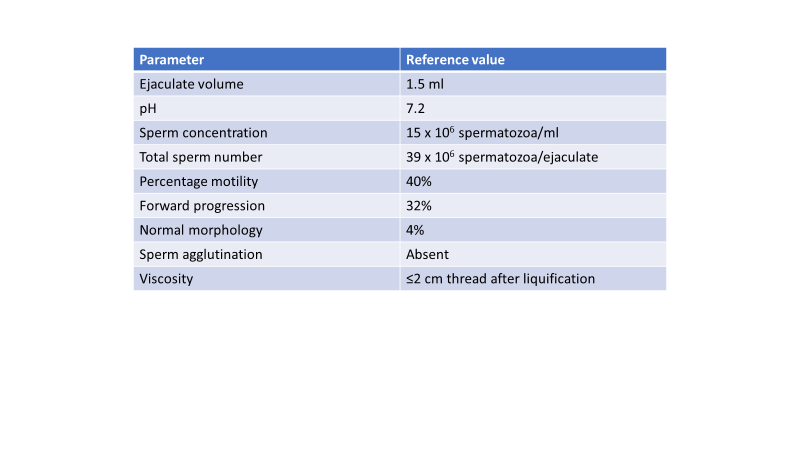
The semen analysis is a snapshot of a male’s semen parameters at that moment in time. Its components include a microscopic assessment of sperm concentration, motility, morphology and the presence of round cells, debris or bacteria. Most REI centers follow World Health Organization (WHO) criteria from 2010 which provides the lower reference limits for each parameter. It’s important to note that a normal semen analysis doesn’t imply fertility and an abnormal semen analysis doesn’t imply subfertility. Historically, the percentage of motile sperm has been reported to correlate best with pregnancy outcomes, but it is now suggested that in order to properly interpret the semen analysis, one must use a combination of parameters as opposed to noting that one or two are low and diagnosing the male with infertility. Although some clinicians prefer two separate specimens obtained 1-3 weeks apart, the more recent consensus seems to be that one specimen is adequate unless the results are abnormal, then another specimen should be obtained with an emphasis on production specifics, such as assuring an ideal abstinence period of 2-5 days, and determining if there was any impediments to producing the sample, such as the male “missing the cup” during the collection process. The subsequent specimen is collected to account for the variability in samples seen within the same individual, not to determine if a treatment or therapy worked.
Most of the components of the semen analysis are self-explanatory, for example, motility describes the percent of sperm that are moving, although there are different levels of sperm progression from twitching in place to actively moving. Morphology refers to a subjective description of how the sperm appear (size and shape) under the microscope. There can be defects in any, and all, of three regions of the sperm cell: the head, neck or tail, which can affect the sperm’s movement or ability to penetrate the oocyte. Low levels of sperm concentration, motility or morphology theoretically imply that the sperm can be impeded in its ability to reach or penetrate the oocyte. Once inside the zona pellucida, though, the sperm should act normally to fertilize and activate the egg, hence the reason why ICSI can result in fertilization in the absence of any normal forms or very low motility.
Lower limits of the accepted reference values for semen analysis
WHO laboratory manual for the examination and processing of human semen, 2010
True to the definition of the word ‘component’ the different values of the semen analysis are just parts of the whole picture, each one not meant to be taken in isolation in order to appreciate an accurate clinical picture of the male patient. The motility and morphology are percentages and the total number of motile or normal sperm, respectively, should be taken into account before a diagnosis of male factor fertility is made. For example, 15 m/ml is the lower limit of concentration and 4% is the lower limit of morphology. If a semen analysis reveals 3% morphology, and the concentration is 15 m/ml that is more concerning than a semen analysis that shows a sample with 3% morphology, but a concentration of 100 m/ml.
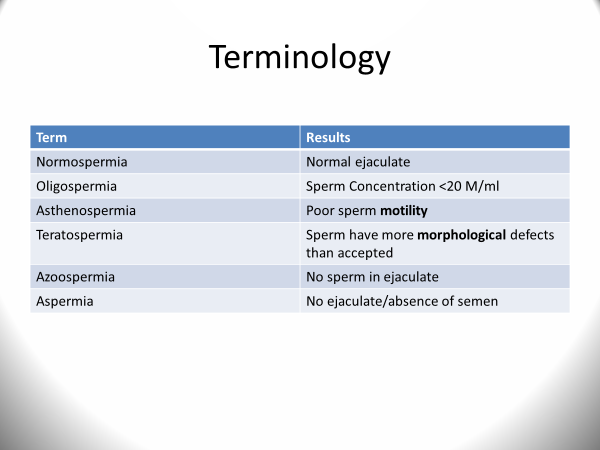
There are some common terms utilized when discussing the interpretation of results. Oligospermia denotes a low sperm count (<15 million/ml) and severe oligospermia is when there is <5 million sperm/ml. Azoospermia is absence of sperm in the ejaculate. Azoospermia is further segregated into two categories: obstructive azoospermia (where sperm is being produced but not delivered, such as a male who had a vasectomy) and non-obstructive azoospermia where sperm is barely being produced if produced at all. In the case of severe oligospermia or non-obstructive azoospermia, it is recommended that a karyotype is obtained on the male to rule out genetic abnormalities. Some experts suggest obtaining a karyotype in cases of oligospermia where the sperm count is <10M/ml based on the fact that chromosomal abnormalities are more common in infertile men than in those who are fertile. Asthenospermia denotes poor sperm motility while teratospermia refers to poor sperm morphology.
Consequently, the semen analysis is an important test but it’s critical to note that it is capturing a biological process at a single moment in time. As a result, there are limitations to its diagnostic and prognostic value. For example, it may show that the amount or shape of sperm are adequate, but it doesn’t assess for the functionality or fertilization potential of those sperm. Also, solely relying on its results can cause one to miss significant conditions or behaviors that can contribute to infertility, such as sexual dysfunction.
Although not yet standards of care, some clinicians utilize additional tests that might uncover damage to sperm that is not revealed in a basic semen analysis, such as DNA fragmentation. DNA integrity is important for normal embryo development. Although more data is needed, a growing body of evidence suggests that high levels of DNA damage can negatively impact reproduction. Sperm samples from infertile men have been shown to have significantly more DNA damage than those from fertile men. Damage to DNA of sperm occurs during their development as well as during the maturation and transport process that takes place outside of the testes. DNA damage can also be due to external factors such as oxidative stress, heat and significant varicocele. The American Society of Reproductive Medicine (ASRM) position The clinical utility of sperm DNA testing states that although some couples may benefit from this testing, it is not recommended for routine use. It also warns that the predictive value for clinical treatment is undetermined and there is no agreed-upon treatment once DNA damage has been diagnosed.
Similarly to unexplained infertility in females, about 30% of men have unexplained or idiopathic infertility as clinicians are unable to determine an apparent cause using the investigative methods described above. Currently, oxidative stress (OS) is believed to be an important and plausible cause of idiopathic male infertility. Simply put, OS reflects an imbalance between reactive oxygen species, which are harmful to the body, and the body’s ability to defend itself (antioxidant defenses). Sperm are susceptible to oxidative stress because they lack the necessary cytoplasmic-enzyme repair systems. OS can result in poor fertilization, poor embryonic development and pregnancy loss. Some examples of OS include poor lifestyle choices such as smoking and excessive alcohol consumption. Others include infection, radiation and high-grade varicoceles. Prevention and management of OS includes diagnosing and treating infection; minimizing detrimental behavior (such as the lifestyle choices listed above), limiting exposure to heat, toxins and heavy metals and ensuring the consumption of anti-oxidants in one’s diet. Additional studies are needed to elucidate the effects of taking anti-oxidants as supplements. Some studies suggest that they may increase sperm parameters, while others find that the data or study design is limited.
Psychological Effects of Infertility on Men
Certainly, whenever communicating with patients, their perspective should be taken into account when delivering results and reviewing and discussing semen analysis results is no exception. Consider the following unique characteristics of men in an infertility setting. They often feel excluded or minimized during the fertility process, even their own lab results are usually reported to their female partner. In addition to the emotional stress that is intrinsic to fertility treatments, men also share the burden of the financial aspect of fertility treatments, which can be costly and can deteriorate the couple’s savings over time. Men in an infertility setting often feel like a failure. The inability to impregnate their partner can be emasculating, regardless of if the fertility factor is male, female or both, which can lead to a compromised sense of identity.
The fertility process itself, and repeated negative outcomes, can lead to depression and anxiety in men as well as women. True to the nature of fertility, even the studies available that focus on the mental health of the male partner in an infertile couple are disproportionately low compared to those of the female partner, but the few that exist confirm that mental health in men is compromised as a result of infertility. A recent one found that 15% of male partners who did not have a successful pregnancy during the study interval met the criteria for being depressed Another group of researchers studied 274 infertile men undergoing treatment and found that 32% were diagnosed with depression and that more than 60% met the clinical criteria for anxiety. Also consider that the couple’s relationship with each other can fall prey to the stress of infertility, particularly if a pregnancy is never achieved. According to one study couples for whom fertility treatments failed were three times more likely to divorce than those who had a child. In addition, the stress of infertility can lead to sexual health problems in the male, like erectile dysfunction, which can hinder treatments, and cause considerable relationship discord, particularly if he is unable to produce a sperm sample on command.
So, what can be done? Researchers recommend that health care providers actively involve male partners in the treatment process. Educating themselves on the male perspective is a valuable first step. A plan should be made in advance regarding how, when and to whom the couple would like male results to be delivered. Realize that men communicate differently and that the male partner may not be as familiar with the clinic’s systems and protocols, for example he may not know the password to access a patient portal in order to obtain his results and must rely on his female partner to do so. One study found that men prefer oral to written communication, but realize that similarly to the female, the male partner is most likely at his job, and can’t listen actively or speak freely. It would be preferable to set up a time in advance to call, maybe during his lunch hour or on his way home from work, if possible. Consider offering mental health support to male partners, but do not be surprised if it’s declined. This same study found that men prefer to have support from infertility clinicians as opposed to mental health professionals.
Male factor infertility is a prevalent issue among infertile couples. The diagnosis can be complex, but at the very least, should include a comprehensive medical history and semen analysis. The semen analysis, although a useful tool, should be interpreted carefully and comprehensively, and in consideration of the entire clinical picture. Other, more specific, tests for male fertility might be useful, but are not yet standard of care. Infertility is stressful, regardless of the cause. Often forgotten, or his presence reduced to only occasional or infrequent contact, the male should be involved in the fertility process whenever possible.
Author
You can visit the Fertility Nurse Newsletter website here
Want to be notified about upcoming Fertility Newsletter posts here at The ObG Project?
All content in this entry was prepared by the above-named author(s) who retain full responsibility for content. The opinions expressed in this article are the author’s own and not those of The ObG Project. Links to third party websites are provided only as a convenience. The ObG Project does not review, control, or examine them. Therefore, The ObG Project is not responsible for the information, advertising, products, resources or other materials of any linked site or any link contained in a linked site. The inclusion of a link does not imply endorsement by the provider. In addition, please be aware that your use of any linked site is subject to the terms and conditions applicable to that site. Please direct any questions regarding linked sites to the webmaster of that site.
Are you an
ObG Insider?
Get specially curated clinical summaries delivered to your inbox every week for free