
This activity is intended for healthcare providers delivering care to women and their families.
After completing this activity, the participant should be better able to:
1. Explain the anatomic significance of a cystic hygroma
2. Discuss the association of cystic hygroma and aneuploidy
Estimated time to complete activity: 0.25 hours
Postgraduate Institute for Medicine (PIM) requires faculty, planners, and others in control of educational content to disclose all their financial relationships with ineligible companies. All identified conflicts of interest (COI) are thoroughly vetted and mitigated according to PIM policy. PIM is committed to providing its learners with high quality accredited continuing education activities and related materials that promote improvements or quality in healthcare and not a specific proprietary business interest of an ineligible company.
The PIM planners and others have nothing to disclose. The OBG Project planners and others have nothing to disclose.
Faculty: Susan J. Gross, MD, receives consulting fees from Cradle Genomics, and has financial interest in The ObG Project, Inc.
Planners and Managers: The PIM planners and managers, Trace Hutchison, PharmD, Samantha Mattiucci, PharmD, CHCP, Judi Smelker-Mitchek, MBA, MSN, RN, and Jan Schultz, MSN, RN, CHCP have nothing to disclose.
Fees for participating and receiving CME credit for this activity are as posted on The ObG Project website. During the period from Dec 31 2017 through Jan 25 2023, participants must read the learning objectives and faculty disclosures and study the educational activity.
If you wish to receive acknowledgment for completing this activity, please complete the test and evaluation. Upon registering and successfully completing the test with a score of 100% and the activity evaluation, your certificate will be made available immediately.

In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and The ObG Project. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Postgraduate Institute for Medicine designates this enduring material for a maximum of 0.25 AMA PRA Category 1 Credit(s)™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
The maximum number of hours awarded for this Continuing Nursing Education activity is 0.2 contact hours.
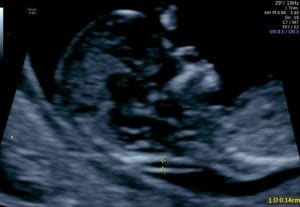
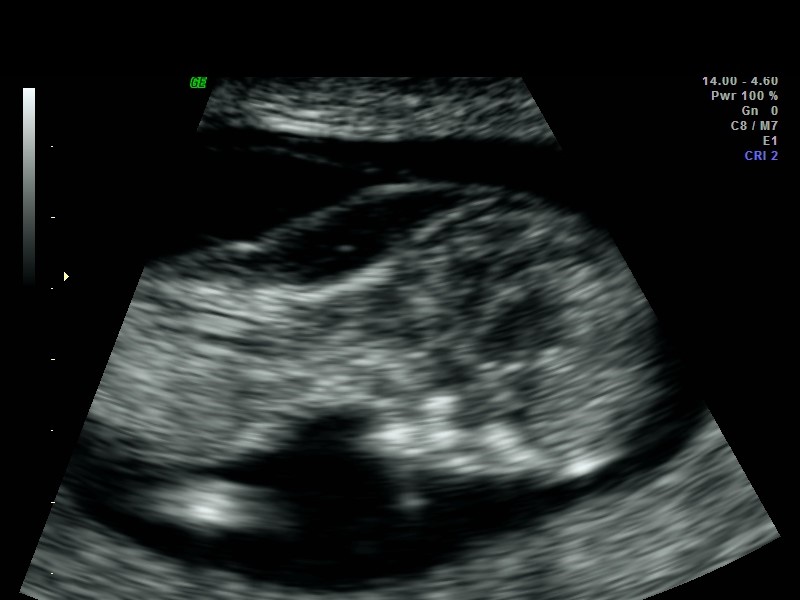
Read Disclaimer & Fine PrintCystic hygroma is a large single or multilocular fluid-filled cavity located in the nuchal region, behind and around the fetal neck, which can extend the length of the fetus and can be seen on fetal ultrasound. Cystic hygroma differs from nuchal translucency (NT). NT is a fluid-filled space normally seen behind the fetal neck on ultrasound performed in the first trimester. Unlike cystic hygroma, NT is a normal structure where size measurement is used as a risk factor for genetic disorders. A thick NT measurement is associated with aneuploidy and other structural anomalies.
A cystic hygroma is an anomaly of the vascular-lymphatic system formation. Failed venous-to-lymphatic connections lead to distended fluid-filled spaces and visualization of septations which may extend along the length of the fetal axis. A cystic hygroma can be seen as early as an 11-week ultrasound. Later in pregnancy, cysts may be seen on either side of the fetal neck, with a nuchal ligament running between the two structures. Aneuploidy is present in 50% of cases. There is a high rate of intrauterine fetal demise especially if hydrops is seen. However, 10 to 20% may resolve in utero in euploid fetuses.
ACOG Practice Bulletin No. 162: Prenatal Diagnostic Testing for Genetic Disorders
ACOG Practice Bulletin 226: Screening for Fetal Chromosomal Abnormalities
First trimester cystic hygroma: does early detection matter?
Maternal Fetal Medicine Specialist Locator-SMFM
Take a post-test and get CME credits
TAKE THE POST TESTThe ObG Project offers links to other third party websites that may be of interest to our website visitors. The links provided in our website are provided solely for your convenience and may assist you in locating other useful information on the Internet. When you click on these links you will leave The ObG Project’s website and will be redirected to another site. These sites are not under the control of The ObG Project.
The ObG Project is not responsible for the software or content of linked third party websites.
Also, please be aware that the security and privacy policies on these sites may be different than The ObG Project’s policies, so please read third party privacy and security policies closely.
If you have any questions or concerns about the products and services offered on linked third party websites, please contact the third party directly.
Are you an
ObG Insider?
Get specially curated clinical summaries delivered to your inbox every week for free
Please log in to ObGFirst to access this page
OBG Project CME requires a modern web browser (Internet Explorer 10+, Mozilla Firefox, Apple Safari, Google Chrome, Microsoft Edge). Certain educational activities may require additional software to view multimedia, presentation, or printable versions of their content. These activities will be marked as such and will provide links to the required software. That software may be: Adobe Flash, Apple QuickTime, Adobe Acrobat, Microsoft PowerPoint, Windows Media Player, or Real Networks Real One Player.
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The planners of this activity do not recommend the use of any agent outside of the labeled indications.
The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of the planners. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information
presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patient’s conditions and possible contraindications and/or dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
It appears you don't have enough CME Hours to take this Post-Test. Feel free to buy additional CME hours or upgrade your current CME subscription plan
You are now leaving the ObG website and on your way to PRIORITY at UCSF, an independent website. Therefore, we are not responsible for the content or availability of this site
